PD-BioStampRC21: Parkinson's Disease Accelerometry Dataset from Five Wearable Sensor Study
- Citation Author(s):
-
Jamie L. Adams (Department of Neurology, University of Rochester Medical Center, Rochester, NY, USA)Karthik Dinesh
(Department of Electrical and Computer Engineering, University of Rochester, Rochester, NY, USA)
Christopher W. Snyder (Rochester General Hospital, Microbiology, Rochester, NY, USA)Mulin Xiong (Michigan State University College of Human Medicine, East Lansing, MI, USA)Christopher G. Tarolli (Department of Neurology, University of Rochester Medical Center, Rochester, NY, USA)Saloni Sharma (Center for Health + Technology, University of Rochester Medical Center, Rochester, NY, USA )E. Ray Dorsey (Department of Neurology, University of Rochester Medical Center, Rochester, NY, USA) - Submitted by:
- Karthik Dinesh
- Last updated:
- DOI:
- 10.21227/g2g8-1503
- Data Format:
 10500 views
10500 views
- Categories:
- Keywords:
Abstract
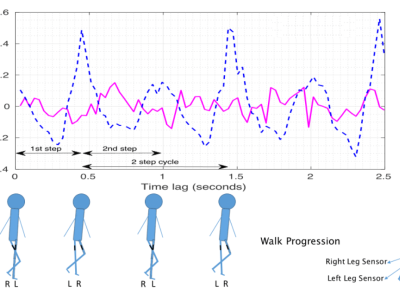
The PD-BioStampRC21 dataset provides data from a wearable sensor
accelerometry study conducted for studying activity, gait, tremor, and
other motor symptoms in individuals with Parkinson's disease (PD). In
addition to individuals with PD, the dataset also includes data for
controls that also went through the same study protocol as the PD
participants. Data were acquired using lightweight MC 10 BioStamp RC
sensors (MC 10 Inc, Lexington, MA), five of which were attached to
each participant for gathering data over a roughly two day
interval.
Instructions:
Users of the dataset should cite the following paper:
Adams JL, Dinesh K, Snyder CW, Xiong M, Tarolli CG, Sharma S, Dorsey E, Sharma G. "A real-world study of wearable sensors in Parkinson’s disease". NPJ Parkinson's disease. 2021 Nov 29;7(1):1-8.
An overview of the study protocol is also provided in the above
mentioned paper. Additional detail specific to the dataset and file
naming conventions is provided here.
The dataset is comprised of two main components: (I) Sensor and
UPDRS-assessment-task annotation data for each participant and (II)
demographic and clinical assessment data for all participants. Each of
these is described in turn below:
I) Sensor and UPDRS-assessment-task annotation data:
The sensor accelerometry and UPDRS-assessment-task annotation data for
all the participants are provided as a zip file named
FullDataSet_PD-BioStampRC21.zip. The size of the zip file is 11GB and,
when unzipped, it generates a set of folders and files with a total
size of approximately 56GB. Unzipping the file generates folders with
name matching the participant ID for each of the Control and PD
participants (17 Control + 17 PD). Each participant folder contains
the data organized as the following files.
a) Accelerometer sensor data files (CSV) corresponding to the five
different sensor placement locations, which are abbreviated as:
1) Trunk (chest) - abbreviated as "ch"
2) Left anterior thigh - abbreviated as "ll"
3) Right anterior thigh - abbreviated as "rl"
4) Left anterior forearm - abbreviated as "lh"
5) Right anterior forearm - abbreviated as "rh"
Example file name for accelerometer sensor data files:
"AbbreviatedSensorLocation"_ID"ParticipantID"Accel.csv
E.g. ch_ID018Accel.csv, ll_ID018Accel.csv, rl_ID018Accel.csv,
lh_ID018Accel.csv, and rh_ID018Accel.csv
File format for the accelerometer sensor data files: Comprises of
four columns that provide a timestamp for each measurement and
corresponding triaxial accelerometry relative to the sensor
coordinate system.
Column 1: "Timestamp (ms)" - Time in milliseconds
Column 2: "Accel X (g)" - Acceleration in X-direction (in units of g = 9.8 m/s^2)
Column 3: "Accel Y (g)" - Acceleration in Y-direction (in units of g = 9.8 m/s^2)
Column 4: "Accel Z (g)" - Acceleration in Z-direction (in units of g = 9.8 m/s^2)
Times and timestamps are consistently reported in units of
milliseconds starting from the instant of the earliest sensor
recording (for the first sensor applied to the participant).
b) Annotation file (CSV). This file provides tagging annotations for
the sensor data that identify, via start and end timestamps, the
durations of various clinical assessments performed in the study.
Example file name for annotation file: AnnotID"ParticipantID".csv
E.g. AnnotID018.csv
File format for the annotation file: Comprises of four columns
Column 1: "Event Type" - List of in-clinic MDS-UPDRS assessments. Each assessment comprises of
two queries - medication status and MDS-UPDRS assessment body locations
Column 2: "Start Timestamp (ms)" - Start timestamp for the MDS-UPDRS assessments
Column 3: "Stop Timestamp (ms)" - Stop timestamp for the MDS-UPDRS assessments
Column 4: "Value" - Responses to the queries in Column 1 - medication status (OFF/ON) and
MDS-UPDRS assessment body locations (E.g. RIGHT HAND, NECK, etc.)
II) Demographic and clinical assessment data
For all participants, the demographic and clinical assessment data are
provided as a zip file "Clinic_DataPDBioStampRCStudy.zip". Unzipping
the file generates a CSV file named Clinic_Data_PD-BioStampRC21.csv
File format for the demographic and clinical assessment data file: Comprises of 19 columns
Column 1: "ID" - Participant ID
Column 2: "Sex" - Participant sex (Male/Female)
Column 3: "Status" - Participant disease status (PD/Control)
Column 4: "Age" - Participant age
Column 5: "updrs_3_17a" - Rest tremor amplitude (RUE - Right Upper Extremity)
Column 6: "updrs_3_17b" - Rest tremor amplitude (LUE - Left Upper Extremity)
Column 7: "updrs_3_17c" - Rest tremor amplitude (RLE - Right Lower Extremity)
Column 8: "updrs_3_17d" - Rest tremor amplitude (LLE - Right Lower Extremity)
Column 9: "updrs_3_17e" - Rest tremor amplitude (Lip/Jaw)
Column 10 - Column 14: "updrs_3_17a_off" - "updrs_3_17e_off" - Rest tremor amplitude during OFF medication assessment
(ordering similar as that from Column 5 to Column 9)
Column 15 - Column 19: "updrs_3_17a_on" - "updrs_3_17e_on" - Rest tremor amplitude during ON medication assessment
Note that columns 10-19 do not contain any data for control
participants and for PD participants that did not participate in the
ON/OFF medication component of the assessment protocol for the study.
For details about different MDS-UPDRS assessments and scoring schemes, the reader is referred to:
Goetz, C. G. et al. Movement Disorder Society-sponsored revision of
the Unified Parkinson's Disease Rating Scale (MDS-UPDRS): scale
presentation and clinimetric testing results. Mov Disord 23,
2129-2170, doi:10.1002/mds.22340 (2008)






In reply to Dear Karthik is there a way by Jiayi Han
In reply to Hello Dear Jiayi, by Karthik Dinesh
In reply to Full dataset is uploaded by Karthik Dinesh